 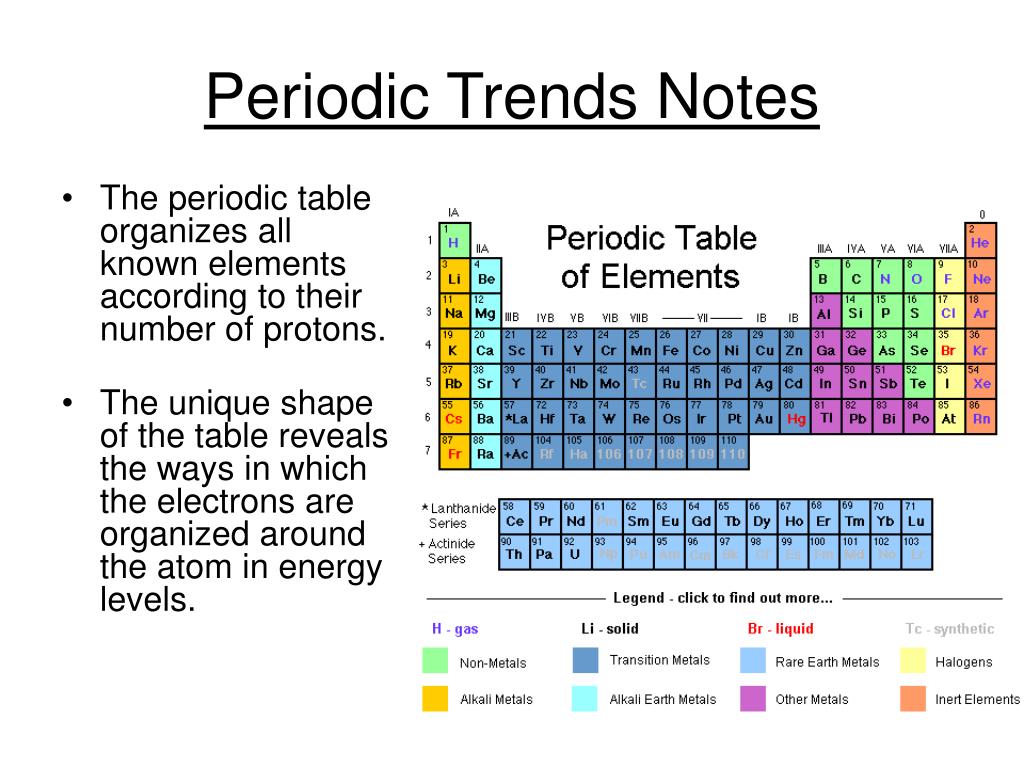
Modern periodic law is the base of periodic trends of properties of elements in the modern periodic table. Periodic Trends of Properties of Elements In Periodic Table In this article, we will discuss periodic properties and their trends in the periodic table in detail. Periodic trends provide chemists with a quick and easy tool to quickly predict the properties of elements. Thus, we can say that elements having similar electronic configurations have similar properties. The recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. The following figure shows the variation of periodic properties of elements.Īlthough we find some exceptions which do not follow these periodic table trends. For example, as we move from left to right in a period, atomic size decreases. That’s why elements show periodicity in their physical and chemical properties in the periodic table. In the modern periodic table elements have been arranged according to their atomic numbers and as stated above atomic numbers are directly related to their physical and chemical properties. Elements can be categorized into s-block, p-block, and d-block elements based on their highest-energy orbital.Moseley gave the Modern Periodic law which states that “Physical and chemical properties of the elements are periodic functions of their atomic numbers”. While there are many orbital shapes that are possible, the MCAT will focus on three particular orbitals denoted as s, p, and d. Some elements, like noble gases, already have a full shell.Įach shell is composed of orbitals: regions of certain shapes in which electrons are likely to be found. Each atom is at its most energetically stable when its valence shell is full of electrons. These electrons are responsible for the reactivity and individual properties of atoms. Valence electrons are electrons on the outermost shell of an atom. This outermost shell is also called the valence shell. The outermost electron shell, or highest-numbered electron shell, is located the furthest. 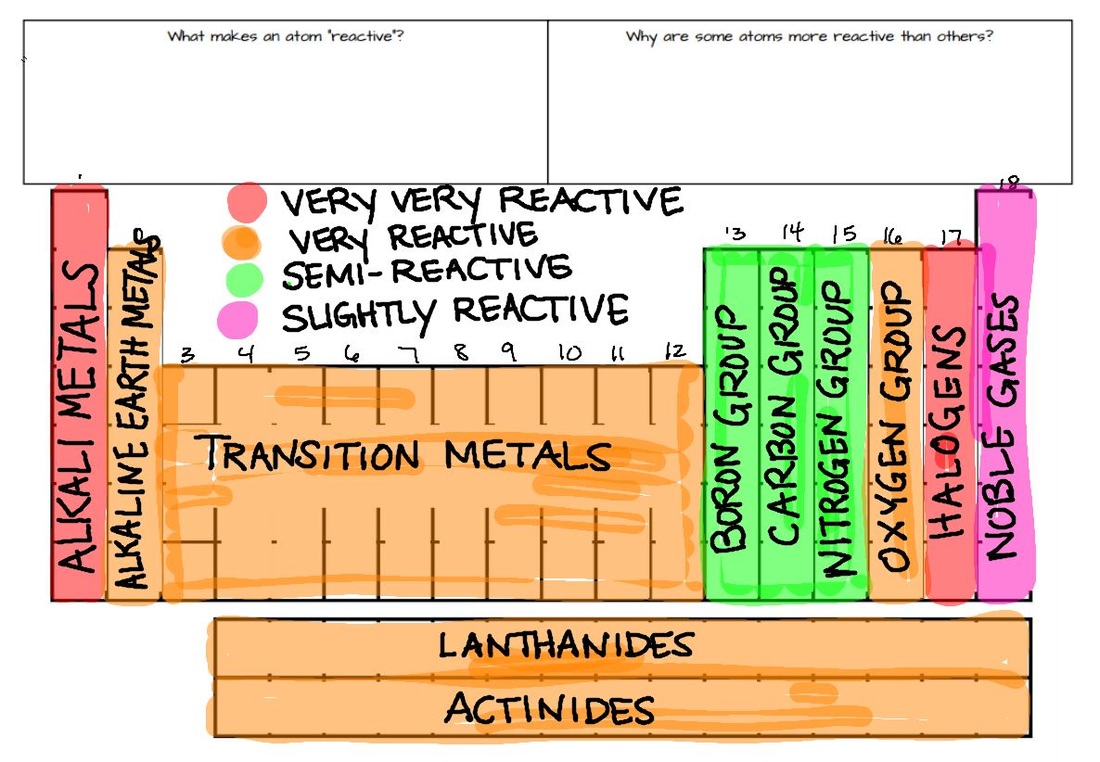
As a general rule, electron shells of a higher number are located further from the nucleus. Thus, elements in the first row of the periodic table have one electron shell, elements in the second row have two electron shells, and so forth. The number of concentric shells is determined by the group number of an atom. While these shells are not physical structures, they are useful in visualizing the spaces that electrons may occupy. When “filling” in an atom, electrons are placed in concentric shells surrounding an atom. For ions, or particles with a net positive or negative charge, the number of electrons may not be equal to the atomic number of the element. Thus, if a particle has zero charge, the number of protons and number of protons must be equal). (Why is this? Recall that the charge of one electron is equal to the charge of one proton. For most atoms, or neutrally charged particles, this number of electrons is equal to the atomic number of the element. Valence electrons play an important role in chemical reactions and bonding.Īn electron configuration is written by determining the number of electrons that are present in the species at hand. The electrons in the outer shell of an atom are called valence electrons. Electrons can be arranged in shells around an atom. In other words, when comparing the mass of a proton or neutron to an electron, the mass of an electron is negligible. Groups of protons and neutrons-collectively referred to as nucleons-form the nucleus of an atom.Īn electron is a subatomic particle that contains a negative charge (-1 e) and has a mass that is 1823x smaller than that of a proton, or 1/1823 atomic mass units. A neutron is a subatomic particle that contains no charge and has a mass of one AMU. “e” refers to the fundamental unit of charge and one atomic mass unit (AMU). A proton is a subatomic particle that contains a positive charge (+1 e). Protons, neutrons, and electrons are the subatomic particles that make up atoms. This is the result of the structure of individual atoms, which are composed of varying subatomic particles. Atoms are the smallest unit of matter that can retain a unique identity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
